The future of biopharma is being shaped by powerful trends in where and how companies invest. But “life sciences” is not a single industry. It’s a collection of highly specialized subsectors, each with its own demands on talent, infrastructure, and location. To understand where the next wave of projects will land, it’s essential to look closer at one of the main subsectors leading the way.
Why Therapeutic Biologics Matter
Therapeutic biologics represent one of the fastest-growing and most strategically important sectors in modern manufacturing. Beyond their role in advancing healthcare, biologics facilities serve as economic anchors.
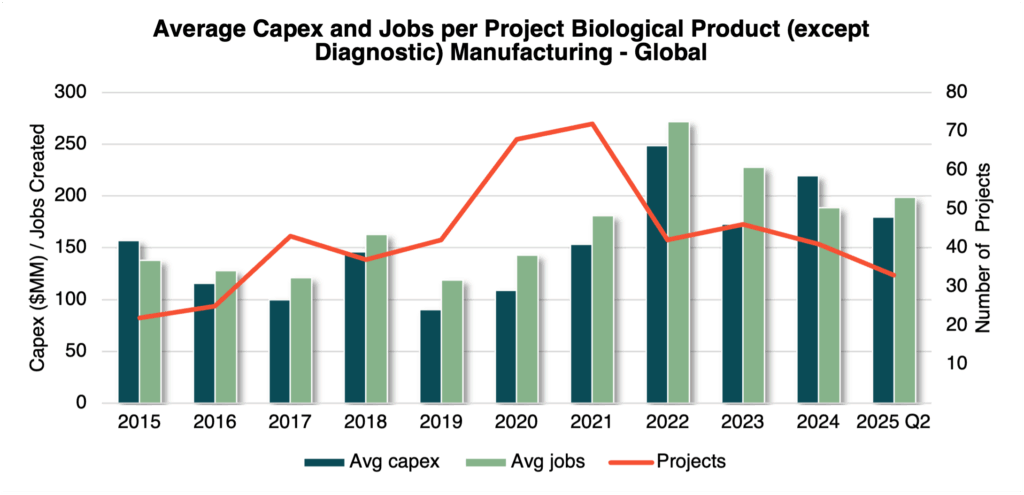
Globally, biologics manufacturing projects now average $180 million in investment and 200 jobs. What was once considered a marquee announcement is quickly becoming the industry norm.
Unlike small-molecule drugs, which rely on chemical synthesis, biologics are produced using genetically engineered cells under highly controlled conditions.
This means facilities must meet stringent standards and are rarely relocated once established. Each site becomes a long-term industrial cornerstone that supports innovation ecosystems and high-value STEM employment.
The COVID-19 pandemic exposed how critical this domestic capacity is. Nations without sufficient biologics manufacturing infrastructure faced severe delays in vaccine and therapy access. A critical reminder that this industry is not only about health but also national preparedness and supply chain security.
Global Trends in Biopharma
Globally, biologics manufacturing has demonstrated significant volatility over the past decade, marked by a sharp surge in 2020–2021 as companies raced to scale up COVID-19 vaccine production through “at-risk” investments. Although activity has since declined, it has stabilized at levels comparable to 2017–2019, indicating a return to steady, sustainable growth rather than a long-term contraction.
At the same time, average project size has increased, reflecting ongoing investment in large-scale, high-impact facilities capable of supporting next-generation therapies. The complexity of biologics production and the requirement for validated facilities tied to specific FDA or EMA approvals, means each site must deliver lasting operational and strategic value.
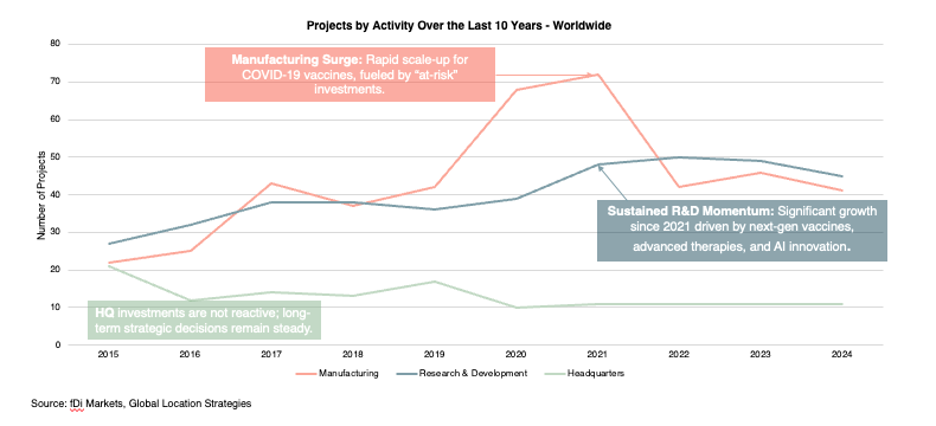
The chart below shows how activity has shifted across three types of projects worldwide over the last decade:
- Manufacturing spiked sharply in 2020–2021 as companies scaled up COVID-19 vaccine production. That surge has since normalized, with project counts declining.
- R&D has shown consistent growth since 2021, reflecting long-term investment in advanced therapies and AI-driven innovation.
- Headquarters projects have remained steady, reflecting the longer-term and less reactive nature of corporate strategy.
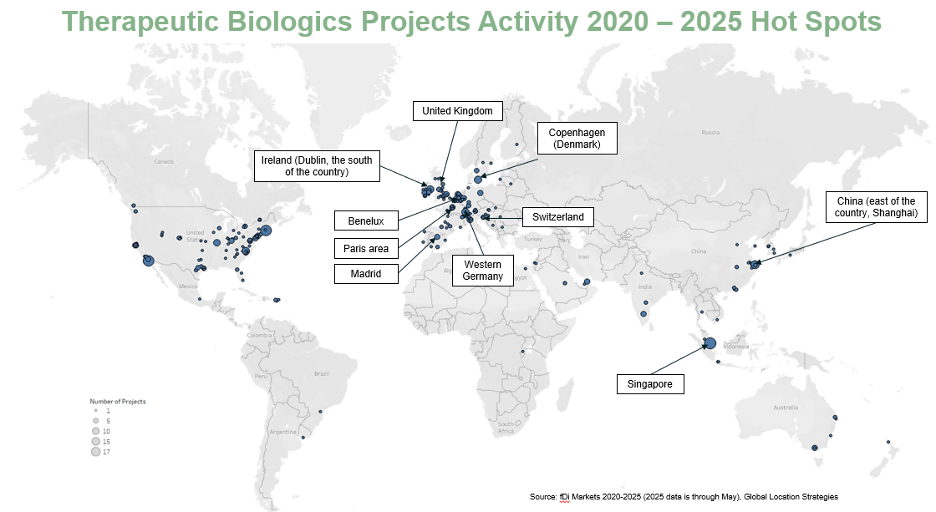
Regionally, Europe has captured the largest share of FDI in the industry over the past decade, averaging roughly 45% of global activity.
The United States accounts for about 28% and remains the second-largest destination.
China has emerged as a leading foreign investor in therapeutic biologics R&D in the U.S. Announcing 12 projects, or 23% of all foreign R&D activity in the U.S, accounting for 50% of China’s total FDI R&D investments in this industry.
The Role of Clusters in Biopharma
Life sciences projects are rarely built in isolation. Companies overwhelmingly favor mature clusters with the right mix of specialized talent, research institutions, and supplier networks.
- Europe currently attracts the largest share of therapeutic biologics manufacturing FDI (about 45% of global projects over the last decade) led by Ireland, which continues to draw the highest number of projects.
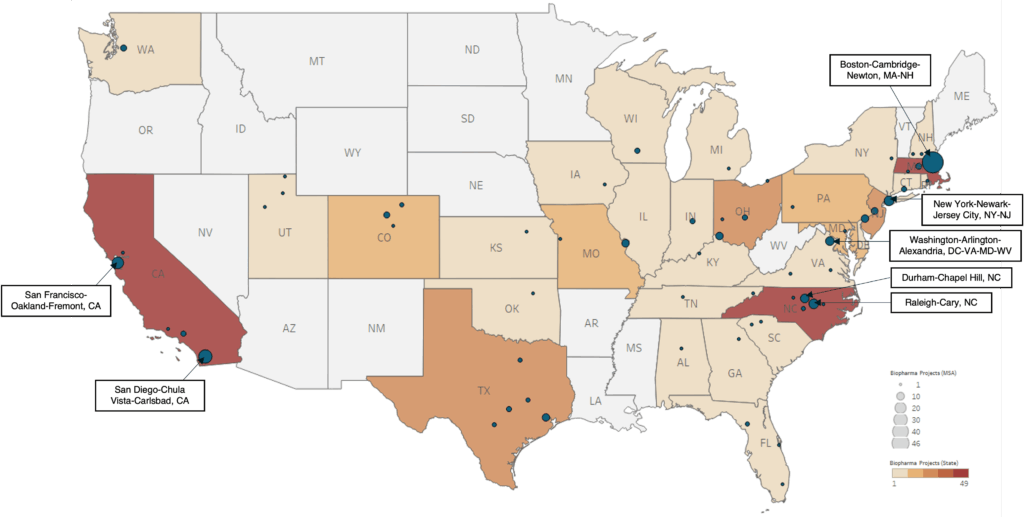
- The United States is the world’s largest biologics market, anchored by powerhouse clusters in Boston, San Diego, San Francisco, and Raleigh–Durham.
- Germany leads as Europe’s top source of outbound investment into the U.S. sector.
- Japan and select parts of Asia round out the global leaders, though most investment activity continues to concentrate in North America and Europe.
Why clusters matter:
- Talent pipelines – Proximity to universities and established industry attracts the bioprocess engineers, regulatory experts, and technicians companies need.
- Supply chain resilience – Mature hubs offer access to vendors, logistics networks, and raw materials, reducing risk in complex operations.
- Innovation advantage – Clusters concentrate R&D activity, making tech transfer and collaboration faster.
- Risk mitigation – Companies see clusters as “safer bets” for high-cost, high-profile projects.
U.S. Specific Trends in Biopharma
The U.S. remains the world largest biologics market, but the nature of those investment is evolving:
- Fewer projects, higher impact – Since 2020, 126 therapeutic biologics manufacturing projects were announced across the U.S., averaging $234 million in CAPEX and 225 jobs per facility. While overall activity has slowed since the pandemic surge, the scale and complexity of each new investment are higher than ever, reflecting growing demand for domestic resilience and onshore production capacity.
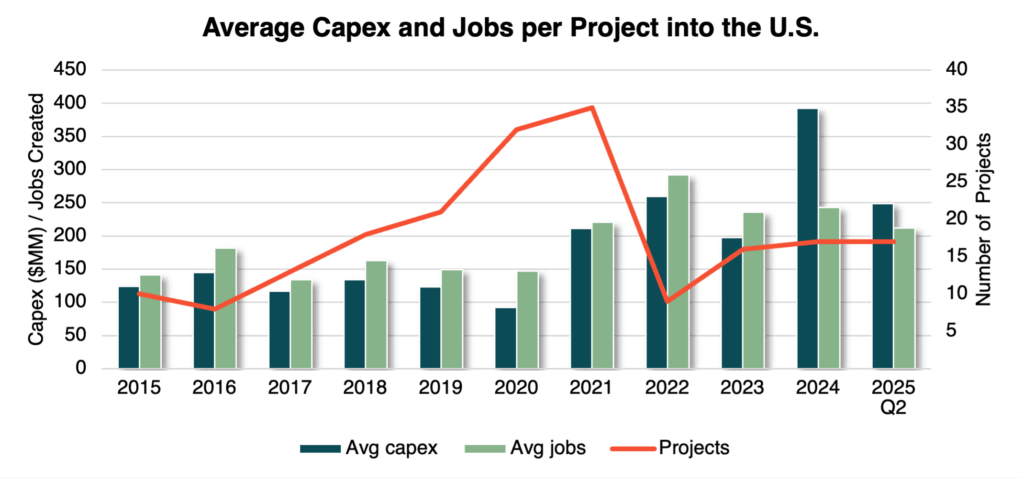
- Geographic concentration – Investment remains anchored in traditional strongholds like California, North Carolina, and Massachusetts, where talent, R&D institutions, and mature supply chains are well established.
At the same time, a new tier of metros is beginning to capture meaningful activity. Markets such as Cincinnati and St. Louis are capturing meaningful biologics activity, signaling a new wave of biologics growth.
The Outlook: Growth, Readiness and Strategic Urgency
The industry’s growth trajectory now depends on readiness. Communities that can align workforce development, infrastructure investment, and regulatory efficiency will be best positioned to secure the next generation of projects.
For companies, this means making location decisions with both speed and strategy. Identifying sites that combine permit-readiness, specialized labor availability, and proven supplier ecosystems.
For regions, it means building partnerships between public and private sectors to close talent gaps, streamline permitting, and market cluster advantages effectively.
The biologics industry is entering a defining moment that will determine not only where the next facilities are built, but who leads in the global race for innovation, health security, and economic resilience.
